- Blog
- Minecraft ddos attack tool
- Mathematica 7 yosemite
- Mac os high sierra 10-13-6 requirements
- Kung ako na lang sana piano sheet music
- Red underlined icon textbar
- Adobe acrobat xi pro upgrade download
- Da lite fast fold vs-bakflip
- How does phonerescue work
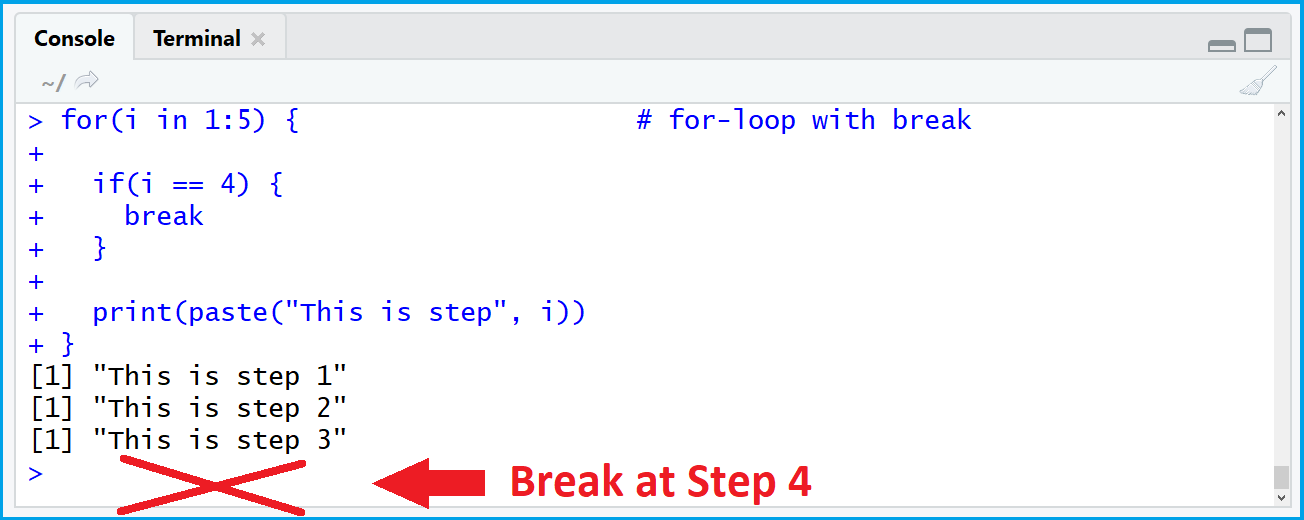
- How to use loop in r
- Lonely planet india travel guide pdf
- Download microsoft office portable 2010
- Mac fonelab how to recovery
- Autoclicker descargar
- Download corel draw 2019
- Red dead redemption wiki

The S9.6 antibody recognizes RNA-DNA hybrids of varying lengths, even those that are only 8 nucleotides long 33. We also incorporated RNase T1 that cleaves single-stranded RNA at guanine 29, 30, and RNase III that cleaves double-stranded RNA including stem-loops 31, 32 to check for nonspecific signals. Our protocol uses the S9.6 antibody 27 to identify RNA-DNA hybrids in R-loops and incorporates RNase H, an endoribonuclease that cleaves and therefore leads to the degradation of the RNA in an RNA-DNA hybrid 20, 28, to ensure that the detected signals are those of hybrids. Here, we describe our assay that identifies R-loops specifically it excludes signals from double-stranded (ds) DNA, double-stranded RNA, and single-stranded RNA. As a preliminary step or in clinical settings to evaluate the effects of mutations on R-loops, these dot-blots must provide sensitive and specific results.
Dot-blots provide such an option since they can be carried out without any specific equipment or computational analysis.

These methods are comprehensive, but they are not practical in many clinical settings or as initial assessments since they require expensive equipment and specialized analysis.Ī procedure that can be done uniformly across laboratories in clinical settings is needed. Immunofluorescence techniques have also been used to quantify and localize R-loops 10, 17. Early versions of these sequencing-based methods did not achieve adequate sequence coverage to allow precise quantitation, but rapid improvement in sequencing technologies now allows locus-by-locus R-loop analysis. To monitor the abundance of R-loops, genome-wide methods first enrich for R-loops with the antibody S9.6 8, 23, 24 or with other nucleases 25 including RNase H 10, 26, 27, and then assess the number of enriched R-loops by sequencing. R-loops are regulated by a variety of proteins including RNase H, senataxin, and other helicases that unwind the RNA-DNA hybrids 18, 19, 20, 21, 22. Like many cellular events, R-loop abundance is tightly titrated too many or too few R-loops impact normal cell function 16, 17. They regulate gene expression by affecting transcription at promoters 10, 11, mediating class-switch recombination 12, and facilitating CRISPR-based genome editing 13, 14, 15. R-loops are also in telomeric regions of chromosomes. R-loops are found in gene promoters 7 and termination 5 sites in mRNA, and along ribosomal RNA 8 as well as transfer RNA 9. Given the many opportunities for their formation, R-loops are not rare, and can be found in 3-5% of the human genome 6, depending on the cell's transcription status. Additionally, R-loops can form when replication and transcription complexes collide 4, and in antisense transcription 5. Outside of the transcriptional complex, the nascent RNA is close to its DNA template, which is still slightly unwound from being copied, thus the RNA can rehybridize with its template DNA forming R-loops 3.
#How to use loop in r free#
The short RNA-DNA hybrid (<10 bp) is resolved to free the nascent RNA so it can leave the RNA polymerase complex through the exit channel 1, 2. In the transcriptional complex, the nascent RNA is synthesized complementary to the template DNA, and the non-template strand is displaced. R-loops are found in different stages of the lifecycle of RNA. R-loop forms when RNA invades a double-stranded DNA to generate an RNA-DNA hybrid and displaces the other DNA strand. This protocol provides a step-by-step guide to a dot-blot assay that allows a quick comparative assessment of the abundance of R-loop, a three-stranded nucleic acid structure. This assay can be used in research and clinical settings to quantify R-loops and assess the effect of mutations in genes such as senataxin on R-loop abundance. This method is highly reproducible, uses general laboratory equipment and reagents, and provides results within two days. Here, we use dot-blots with the S9.6 antibody to quantify R-loops and show the sensitivity and specificity of this assay with RNase H, RNase T1, and RNase III that cleave RNA-DNA hybrids, single-stranded RNA, and double-stranded RNA, respectively. A challenge in the field is the quantitation of R-loops since much of the work relies on the S9.6 monoclonal antibody whose specificity for RNA-DNA hybrids has been questioned. Next, it is critical to understand the roles of R-loops and how cells balance their abundance. Initially, R-loops were thought to be the by-products of transcription but recent findings of fewer R-loops in diseased cells made it clear that R-loops have functional roles in a variety of human cells. The three-stranded nucleic acid structure, R-loop, is increasingly recognized for its role in gene regulation.
- Blog
- Minecraft ddos attack tool
- Mathematica 7 yosemite
- Mac os high sierra 10-13-6 requirements
- Kung ako na lang sana piano sheet music
- Red underlined icon textbar
- Adobe acrobat xi pro upgrade download
- Da lite fast fold vs-bakflip
- How does phonerescue work
- How to use loop in r
- Lonely planet india travel guide pdf
- Download microsoft office portable 2010
- Mac fonelab how to recovery
- Autoclicker descargar
- Download corel draw 2019
- Red dead redemption wiki
